White House officials have a plan to ramp up the availability of Pfizer’s COVID-19 antiviral Paxlovid, according to a report from Bloomberg. White House Covid-19 Response Coordinator Dr. Ashish Jha said that the administration would announce more details about the plans to make Paxlovid more widely available in the coming week. The administration plans to launch…
Johnson & Johnson rises on mixed bag Q1, increased 2022 guidance
Johnson & Johnson (NYSE:JNJ) shares ticked up today on first-quarter results that were mixed compared to the consensus forecast. The New Brunswick, New Jersey-based company posted profits of $5.1 billion, or $1.93 per share, on sales of $23.4 billion in the first quarter for a 16.9% bottom-line slide on sales growth of nearly 5%. Get the…
Developing nations shun AstraZeneca’s COVID-19 vaccine
A number of developing nations have refused tens of millions of doses of AstraZeneca’s COVID-19 vaccines, according to a report from Reuters. The countries had received doses of the AstraZeneca vaccine from the COVAX initiative, a global project that focuses on ensuring equitable access to COVID-19 vaccines. COVAX has distributed 1.4 billion shots to 144 nations…
NIH starts evaluating second COVID-19 booster shots in adults
The NIH recently announced that it has started enrolling adults for a Phase 2 clinical trial evaluating various additional COVID-19 booster shots. NIH’s National Institute of Allergy and Infectious Diseases (NIAID) is sponsoring the COVID-19 Variant Immunologic Landscape (COVAIL) trial. “We are looking beyond the Omicron variant to determine the best strategy to protect against…
FDA backs second COVID-19 vaccine booster for some Americans
FDA has authorized a second booster dose of COVID-19 vaccine for individuals who are immunocompromised or 50 and over. The expanded emergency use authorization applies to the vaccines from Pfizer (NYSE: Pfizer) and Moderna (Nasdaq: MRNA). Individuals interested in obtaining a second booster dose must wait at least four months after receiving the first booster dose.…
FDA advisory panel to convene to consider second booster dose
The Vaccines and Related Biological Products Advisory Committee (VRBPAC) is scheduled to meet on April 6 to discuss the potential use of COVID-19 vaccine booster dose. Both Pfizer (NYSE:PFE) and Moderna (Nasdaq:MRNA) are seeking to amend the emergency use authorization of their respective vaccines to allow for an additional booster. Pfizer and its partner BioNTech SE (Nasdaq:BNTX)…
Manufacturers ink deal to make generic version of Pfizer’s oral COVID-19 pill Paxlovid
The Medicines Patent Pool (MPP) announced that it has signed agreements with 35 manufacturers to make an inexpensive version of Pfizer’s (NYSE: PFE) Paxlovid (nirmatrelvir/ritonavir) to be distributed in 95 low- and middle-income countries. Vaccination rates in many developing countries lag far behind that of wealthier countries. In Nigeria, for instance, 9.1% of the population…
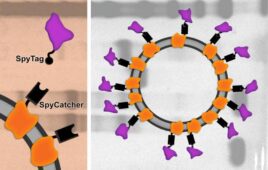
Engineered yeast could be key for more affordable COVID-19 vaccines
Researchers at MIT and Beth Israel Deaconess Medical Center are exploring a yeast-based alternative to RNA vaccines. A new paper highlights a vaccine, which comprises fragments of the SARS-CoV-2 spike protein arrayed on a virus-like particle, that reportedly elicited a strong immune response and protected animals against viral challenge, according to a post on MIT’s…
A closer look at Honeywell’s and Walvax Biotech’s mRNA COVID-19 vaccine plant partnership
Walvax (SHE: 300142) recently formed a strategic collaboration with Honeywell (Nasdaq: HON) that led to the construction of China’s first mRNA COVID-19 vaccine production plant. The company has been working since 2017 to become a leading vaccine R&D company, recounted Shawn Opatka, vice president, life sciences, Honeywell Process Solutions. “Their goals accelerated the construction of…
Sanofi details €2B plan to make France an mRNA leader
French drugmaker Sanofi (Nasdaq:SNY) said it would spend €935 million (about $1.1 billion) between 2022 and 2026 to produce mRNA-based vaccines. That funding is part of a larger €2 billion (about $2.4 billion) initiative to accelerate its mRNA development capability, Sanofi explained on its French-language website. The company intends to use the funds to ramp…