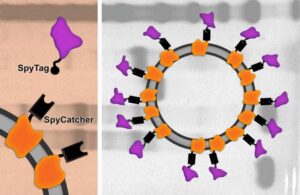
A new protein subunit vaccine developed at MIT and Beth Israel Deaconess Medical Center may offer an inexpensive, easy-to-store, and effective alternative to RNA vaccines for COVID-19. Pictured is a schematic of the vaccine.
[Image from Jose-Luis Olivares, MIT, and figures courtesy of the researchers]
A new paper highlights a vaccine, which comprises fragments of the SARS-CoV-2 spike protein arrayed on a virus-like particle, that reportedly elicited a strong immune response and protected animals against viral challenge, according to a post on MIT’s website.
The researchers designed the vaccine to be produced by yeast, using fermentation facilities that already exist around the world. MIT said the Serum Institute of India — the world’s largest vaccine manufacturer — is now producing large quantities of the vaccine and plans to run a clinical trial in Africa.
According to the researchers, protein-based, subunit vaccines are low-cost and can provide a consistent supply in parts of the world that does not have access to vaccines, namely the ones protecting against COVID-19.
MIT professor of chemical engineering J. Christopher Love and BIDMC Director for the Center of Virology and Vaccine Research Dan Barouch are the senior authors of the paper supporting the vaccine, which was published in Science Advances. MIT graduate students Neil Dalvie and Sergio Rodriguez-Aponte, and Lisa Tostanoski, a postdoc at BIDMC, are lead authors of the paper.
The researchers aimed to produce a vaccine that would be both effective and easy to manufacture, focusing on protein subunit vaccines that consist of small pieces of viral proteins and can often be stored under refrigeration rather than the ultracold storage required by RNA vaccines.
In the subunit vaccine, the researchers used a small piece of the SARS-CoV-2 spike protein, the receptor-binding domain (RBD), and to make it more immunogenic, they displayed copies of the protein on the virus-like particle. When coated with the SARS-CoV-2 RBD fragments, the particle produced a much stronger response than the RBD protein on its own.
To ensure the vaccine could be manufactured easily and efficiently, the researchers designed the RBD protein so it could be produced by the yeast Pichia pastoris, which is relatively easy to grow in an industrial bioreactor, according to MIT. That yeast has already been used to produce vaccines in bioreactors around the world.
“One of the key things that separates our vaccine from other vaccines is that the facilities to manufacture vaccines in these yeast organisms already exist in parts of the world where the vaccines are still most needed today,” Dalvie said on the MIT post.
Testing occurred in non-human primates, demonstrating that the vaccine generated antibody levels similar to those produced by some of the approved COVID-19 vaccines, including Johnson & Johnson’s. Viral loads in vaccinated animals were lower than those seen in unvaccinated animals when exposed to SARS-CoV-2 as well, the researchers said.
Additionally, the researchers have incorporated two mutations similar to those identified in the delta and lambda variants as they say their approach offers a “plug and display” system that could create similar vaccines.
“We could make mutations that were seen in some of the new variants, add them to the RBD but keep the whole framework the same, and make new vaccine candidates,” Rodriguez-Aponte said. “That shows the modularity of the process and how efficiently you can edit and make new candidates.”
Researchers from the Serum Institute and SpyBiotech also contributed to the paper. The research was funded by the Bill and Melinda Gates Foundation and the Koch Institute Support (core) Grant from the National Cancer Institute.




Tell Us What You Think!