Purdue Pharma and the Sackler family will pay $6 billion to settle lawsuits with a host of states over their role in the opioid epidemic. Purdue Pharma and the Sackler family — the founders and owners of the company — will pay the $6 billion to victims, survivors and states as a result of their…
Ember Technologies, Cardinal Health partner on self-refrigerated, cloud-based shipping box
Ember Technologies and Cardinal Health (NYSE:CAH) announced that they are partnering on the Ember Cube digital shipping box. The two companies will collaborate to offer what they are touting as the world’s first self-refrigerated, cloud-based shipping box, according to a news release. They will work together to deliver a cold chain solution designed to ensure product integrity and…
DHL invests $400M in its pharma and medtech supply chain
DHL Supply Chain announced today that it will invest upwards of $400 million to expand its pharmaceutical and medical device distribution network footprint. Westerville, Ohio-based DHL Supply Chain’s life sciences and healthcare (LSHC) sector will make the investment to expand the supply chain footprint by 27% this year, amounting to nearly 3 million additional square…
Cardinal Health to pay $13M to settle claims it paid kickbacks to doctors
The U.S. Department of Justice announced that Cardinal Health (NYSE:CAH) agreed to pay more than $13 million to resolve kickback allegations. Allegations against Dublin, Ohio-based Cardinal Health include a violation of the False Claims Act by paying “upfront discounts” to its physician practice customers in violation of the Anti-Kickback Statute. The Anti-Kickback Statute prevents pharmaceutical…
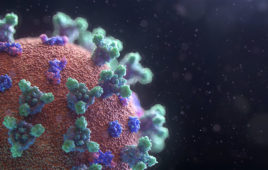
FDA fully approves Moderna’s COVID-19 vaccine
Moderna (NSDQ:MRNA) announced today that it received full FDA approval of the biologics license application for its COVID-19 vaccine. FDA’s approval for the company’s Spikevax mRNA COVID-19 vaccine covers the prevention of the virus in individuals aged 18 years or older. Moderna submitted for full FDA approval back in June 2021 and becomes the second…
B. Braun receives FDA approval for Florida pharmaceutical manufacturing plant
B. Braun Medical announced today that it received final approval from the FDA for its new pharmaceutical manufacturing plant. Bethlehem, Pennsylvania-based B. Braun’s Daytona Beach, Florida-based plant will produce 0.9% sodium chloride for injection available in B. Braun’s Excel Plus IV bags in 1,000 mL and 500 mL sizes. According to a news release, the…
Fujifilm to aquire Atara’s T-cell manufacturing facility in California for $100M
Atara Biotherapeutics announced yesterday that Fujifilm will acquire its T-cell operations and manufacturing (ATOM) facility for $100 million. South San Francisco-based Atara entered into a long-term strategic agreement with Fujifilm’s Diosynth Biotechnologies (FDB) subsidiary under which Fujifilm will acquire the ATOM facility in Thousand Oaks, Calif., and retain current manufacturing and quality staff at the…
Generic manufacturers to make cheaper version of Merck’s COVID-19 pill for developing countries
The Medicines Patent Pool (MPP) announced today that it signed agreements with 27 manufacturers to produce Merck’s COVID-19 pill. MPP’s agreements with the generic manufacturing companies cover the manufacturing of the oral COVID-19 antiviral medication molnupiravir, with supply set for 105 low- and middle-income countries (LMICs). The agreements come as a result of the voluntary…
FDA postpones some inspectional activities amid omicron surge
The FDA announced earlier this week that it made some temporary changes to its inspectional activities due to COVID-19. With the omicron variant of the virus causing surges across the world, the FDA implemented the changes in the U.S. on Dec. 29, 2021, to ensure the safety of its employees and the firms it regulates,…
Fareva, ApiJect ink 10-year licensing agreement on fill-finish production lines
Fareva and ApiJect Systems today announced a licensing agreement to fill-finish more than 500 million injectable drug doses per year. Under the agreement, Fareva intends to invest more than $56.5 million to install three blow-fill-seal production lines with support from the Government of France through the France Relance industry initiative, as well as the Investments for the…