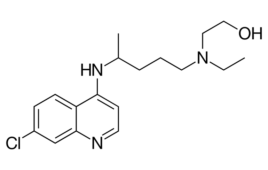
A National Institutes of Health clinical trial has concluded that hydroxychloroquine had no clinical benefit for hospitalized COVID-19 patients. The Journal of the American Medical Association published the results from the blinded, placebo-controlled, randomized trial. The clinical trial, known as the “ORCHID” study, treated 479 symptomatic COVID-19 patients with hydroxychloroquine in 34 hospitals in the…
5 ways Election Day could impact pharma
The pharmaceutical industry stands at a crossroads as Election Day nears in the U.S. The election could ultimately have sweeping implications for the sector, or it could yield only modest changes. While the presidential election could have a significant influence on the pharma sector, arguably more important is who gains control of Congress. A blue sweep…
Report: Top FDA official vows to quit if vaccine approved prematurely
A top FDA official has threatened to resign if the Trump administration moves too quickly to approve a vaccine for COVID-19, according to a published report. Peter Marks, director of the agency’s Center for Biologics Evaluation and Research (CBER), made the threat over concerns a vaccine would be approved without being proven safe and effective,…
Trump signs executive order to help reshore pharmaceutical manufacturing
President Donald Trump recently signed an executive order meant to encourage the reshoring of pharmaceutical manufacturing to the U.S. The executive order, signed Aug. 6, includes “buy American” provisions for U.S. government agencies’ procurement of essential medicines and reduces regulatory provisions. The Secretary of Health and Human Services and FDA commissioner, consistent with applicable law,…