Nostrum Laboratories is initiating the voluntary recall of two lots of its Metformin HCI extended-release tablets for people with diabetes. According to an FDA news release, the Metformin HCI extended-release tablets (USP 750mg) were found to contain levels of nitrosamine impurities above the ADI limit of 96ng per day, as stipulated in the FDA Guidance Document…
Flu vaccination worries rise in South Korea following deaths
Some 72 people have died in South Korea after receiving flu shots, leading several physicians to call for a pause in immunizations. Government officials initially reported there was no clear link between the deaths and the vaccines. Among the casualties was a 17-year-old high school student. The Korea Disease Control and Prevention Agency (KDCA) decided…
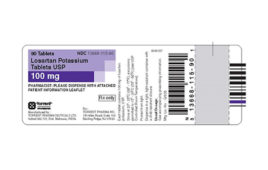
Torrent Pharmaceuticals Issues Voluntary Nationwide Recall of Losartan Potassium Tablets
Torrent Pharmaceuticals Limited is voluntarily recalling two lots of Losartan potassium tablets, USP to the consumer level due to the detection of trace amounts of an unexpected impurity found in an active pharmaceutical ingredient (API) manufactured by Hetero Labs Limited. The impurity detected in the API is N-nitrosodiethylamine (NDEA), which is a substance that occurs…
Latex Hazard Leads to Recall of Certain Dyural-40 and Dyural-80 Convenience Kits
Asclemed USA Inc., a unit of Enovachem Pharmaceuticals, is voluntarily recalling 20 lots of Dyural-40 and 61 lots of Dyural-80, to the user level. The products include recalled Sodium Chloride, USP, 0.9% manufactured by Fresenius Kabi, which has been recalled due to product labeling incorrectly stating stoppers do not contain latex. For the population most at…
Tris Pharma Issues Recall of Infants’ Concentrated Oral Suspension Ibuprofen
Tris Pharma, Inc. has voluntarily recalled three lots of Infants’ Ibuprofen Concentrated Oral Suspension, USP (NSAID) 50 mg per 1.25 mL, to the retail level. The recalled lots of the product have been found to potentially have higher concentrations of ibuprofen. There is a remote possibility that infants, who may be more susceptible to a higher…
Pfizer Issues Voluntary Nationwide Recall of Six Lots of Thermacare Heatwraps
Mylan Initiates Nationwide Recall of 15 Lots of Valsartan Tablets
Mylan Pharmaceuticals is conducting a voluntary nationwide recall of select lots of Valsartan-containing products due to trace amounts of an impurity, N-nitrosodiethylamine (NDEA), contained in the API Valsartan, USP, manufactured by Mylan Laboratories Limited. NDEA is a substance that occurs naturally in certain foods, drinking water, air pollution, and industrial processes, and has been classified as a probable human…
Drug Recalls Increased, Medical Device Recalls Decreased in Q3
In the third quarter of 2018, faulty manufacturing practices had an impact on the pharmaceutical industry, according to the latest Recall Index released November 13 by Stericycle Expert Solutions. Drug recalls increased 19 percent to 92, though recalled units declined 56 percent to just over 7 million; lower than 10 of the last 13 quarters. Failed specifications…
FDA Alerts Consumers of Voluntary Recall of Puriton Eye Relief Drops
The Food and Drug Administration is alerting consumers about a voluntary recall of all lots of Puriton Eye Relief Drops, 0.5 oz. (15 ml) bottle. These products are labeled as a homeopathic and are being recalled due to non-sterile production conditions at the manufacturing facility. Consumers should stop using this product and contact their physician or…
FDA Update: Angiotensin II Receptor Blocker Recalls
FDA is alerting patients and health care professionals to ScieGen’s voluntary recall of certain lots of irbesartan, an angiotensin II receptor blocker (ARB), because they contain N-Nitrosodiethylamine (NDEA), a known animal and suspected human carcinogen (causes cancer). FDA laboratory testing confirmed NDEA in some lots of ScieGen’s irbesartan. ScieGen’s irbesartan products are labeled as Westminster Pharmaceuticals and…