(Credit: FDA MedWatch)
Certain homeopathic teething products: Recall — confirmed elevated levels of belladonna.
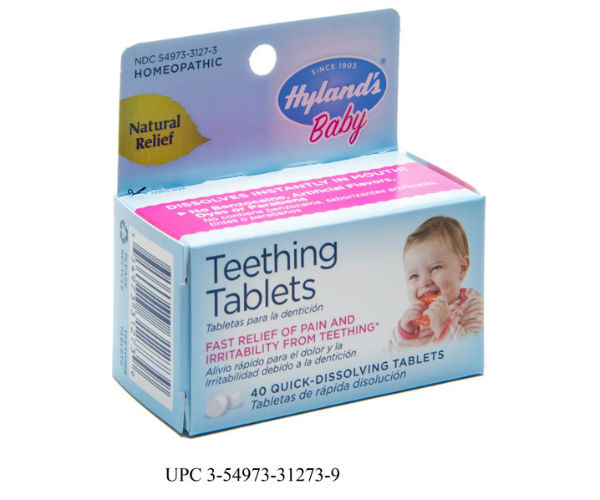
FDA is alerting consumers of a nationwide voluntary recall of all lots of Hyland’s Baby Teething Tablets and Hyland’s Baby Nighttime Teething Tablets by the manufacturer, Standard Homeopathic Company, which is located in Los Angeles. On April 10, the company received FDA’s formal recall request and agreed to conduct a recall.
Standard Homeopathic Company is notifying its distributors and retailers by mail and is arranging for the return of all recalled products. Consumers who have products which are being recalled should contact the company.
Consumers with questions regarding this recall can contact Standard Homeopathic Company by calling 1-800-991-3376 (Monday-Friday 6 a.m. to 4 p.m. Pacific Time). Consumers should contact their physician or healthcare provider if they believe they have experienced any problems that may be related to taking or using this drug product.
FDA reminds consumers that homeopathic teething tablets containing belladonna pose an unnecessary risk to infants and children and urges consumers not to use these products.
ISSUE:
FDA announced that its laboratory analysis found inconsistent amounts of belladonna, a toxic substance, in certain homeopathic teething tablets, sometimes far exceeding the amount claimed on the label. The agency is warning consumers that homeopathic teething tablets containing belladonna pose an unnecessary risk to infants and children and urges consumers not to use these products.
In light of these findings, the FDA contacted Standard Homeopathic Company in Los Angeles, the manufacturer of Hyland’s homeopathic teething products, regarding a recall of its homeopathic teething tablet products labeled as containing belladonna, in order to protect consumers from inconsistent levels of belladonna.
BACKGROUND:
Homeopathic teething products have not been evaluated or approved by the FDA for safety or effectiveness. The agency is unaware of any proven health benefit of the products, which are labeled to relieve teething symptoms in children. In September 2016, the FDA warned against the use of these products after receiving adverse event reports.
In November 2016, Raritan Pharmaceuticals (East Brunswick, New Jersey) recalled three belladonna-containing homeopathic products, two of which were marketed by CVS.
RECOMMENDATION:
The FDA recommends that consumers stop using these products marketed by Hyland’s immediately, and dispose of any in their possession.
Consumers should seek medical care immediately if their child experiences seizures, difficulty breathing, lethargy, excessive sleepiness, muscle weakness, skin flushing, constipation, difficulty urinating, or agitation after using homeopathic teething products.
Healthcare professionals and patients are encouraged to report adverse events or side effects related to the use of these products to the FDA’s MedWatch Safety Information and Adverse Event Reporting Program.
Click here to read the MedWatch safety alert, along with a link to the press release.
Follow us on Twitter and Facebook for updates on the latest pharmaceutical and biopharmaceutical manufacturing news!
(Source: FDA MedWatch)




