[Image courtesy of Benjamin Vincent Kasapoglu via Wikimedia Commons]
Experts remain that patients who have experienced trouble getting prescriptions filled switch to other similar amphetamine-based medications, according to The Washington Post.
Teva (NYSE:TEVA) recently disclosed that it could struggle to supply the drug for the next two to three months. However, the company is “producing and distributing as much as possible, as quickly as possible,” according to Teva spokesperson Kelley Dougherty.
Novartis (NYSE:NVS) subsidiary Sandoz has also acknowledged having a generic Adderall shortage. Sandoz is the second biggest manufacturer of the drug.
Bloomberg reported that Sandoz had supply problems with both the drug’s extended- and immediate-release versions.
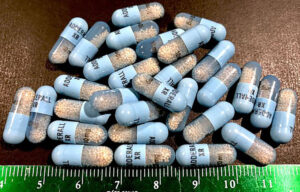
DEA sets annual limits on sales of Adderall and generics each year. Those quotas have contributed to shortages of the drug in the past.
Some patients have had to wait multiple months to have prescriptions filled, according to Generics Bulletin.
Currently, seven out of nine manufacturers of the drug report Adderall shortages. The latest to join the list are Sun Pharma and Camber Pharmaceuticals.
The American Society of Health-System Pharmacists (ASHP) has documented supply problems related to Adderall generics since August.
According to ASHP, affected manufacturers did not provide a reason for the shortage.
In August, Teva reported a labor shortage on the packaging line for its 20-mg and 30-mg Adderall generic tablets.
FDA recently added Adderall generics to its database of drug shortages. The agency acknowledged Teva’s manufacturing delays as contributing to the shortage of the drug.
The FDA also cited an Adderall drug shortage occurring from September 2019 to 2022.
Demand for Adderall, which was first approved in 2001, has increased in recent years. Last year, there were 41 million prescriptions for the drug compared to 21.3 million in 2012, according to estimates from Symphony Health.
Adderall is a Schedule II drug and is commonly abused.
During the pandemic, a crop of Telehealth vendors began to exploit relaxed guidelines restricting mailing scheduled substances. The federal government is reportedly investigating two of those companies — Cerebral and Done — for possible violations of the Controlled Substances Act, according to The Wall Street Journal.
Earlier this year, FDA and DEA sent joint warning letters to owners of the websites kubapharm.com and premiumlightssupplier.com that claimed to sell Adderall and other medicines without a prescription.
This article was updated on October 13 with new information.





My local CVS pharmacy gave my medication that I pick up the same time every month to someone else I really depend on this for my part-time job after being diagnosed five years ago following stage for cancer I’m worried I have anxiety that I’m going to get in trouble with my marriage lose my job and the local police are extremely brutal, and lacking in any mental health education just brutal. Boynton Beach Florida
What really needs to be discussed here is the importance of prescribers providing patients with 90 day and or 120 day supply of this life saving medication. It is irresponsible as hell to not provide large quantities to PREVENT CODEPENDENCY AND PREVENT INCREASED COSTS that occur with month to month refills.
It has always been the same cost IF NOT CHEAPER to purchase 90 day and 120 day quantities of this medication AND ANY MEDICATION, for that matter.
My insurance REQUIRES my prescribers to provide at the minimum 90 day refills, lest they be forced to provide a damn good reason as to why they can’t.
I welcome any medication maker, etc to contact me as id love to be part of any commercials or news media advocating the need of this life saving medicine!!!
The framing of this issue as due to “overprescription” by companies whose goal is to get more people diagnosed is disingenuous and dangerous. ADHD meds are some of the most effective mental health intervention with the least side effects in the history of medicine, no exaggeration, and there is no “right” number of prescriptions or diagnoses. Taking ADHD medications and having them make a patient’s life better *is itself diagnostic.* People who don’t have the right brain chemistry find these meds highly unpleasant and don’t tolerate long-term use, and generally discontinue it on their own in short order.
But moralizing over “college kids” has made these medications highly gate-kept and very hard to get for those who have a life-impacting disability and makes it hard to get life-altering treatment. This also means it’s been hard to get a diagnosis; but COVID helped, in a way. It meant more people had disruptions in their lives that their coping mechanisms couldn’t handle, and they sought help — and telemedicine finally happened universally, making that help easier to get. It didn’t mean they didn’t have ADHD before!
Unfortunately the DEA regulates how much can get made, and thus every year there is a shortage. This year is the worst, but this *isn’t new*. Even though the likelihood of abuse is small, and the impact of a shortage on patients is astronomical, the DEA has decided there is a “right” number of people who have ADHD and somehow they know what that number is.
In the meantime, people with disabilities get hurt. Please consider your reporting through the lense of those very real people, and not just anti-psychiatric-medicine sensationalism.